Research Interest
2. Nano-/Micro-scale Thermal-Fluid Transport and Conversion
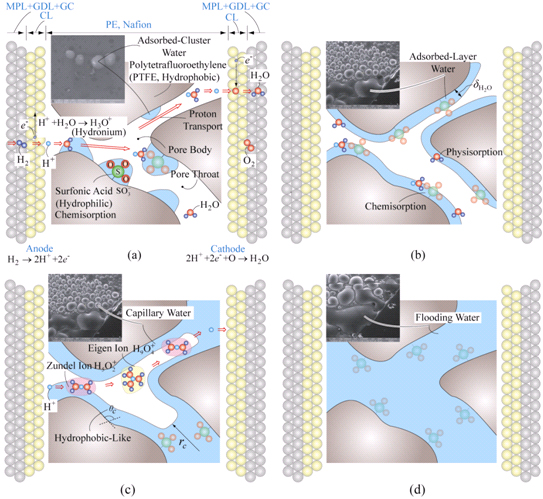
3. Nanoscale Thermal Management System
When gas is confined in spacing with characteristic length L comparable or smaller than its mean free path λf (average travelled distance without collision with adjacent particles), the effective thermal conductivity of the gas is reduced due to interaction of the gas particles with the confining surfaces. Condensable atoms/molecules are also adsorbed to the surfaces by interatomic/molecular potential near saturation temperature and this adsorbed layer influences energy and momentum exchange, i.e., the thermal aT and momentum au accommodation coefficients.
Effect of adsorption on momentum au and thermal aT accommodation coefficients and effective thermal conductivity of Ar vapor-filled Pt nanogap and nanocavity is examined using nonequilibrium MD. For the accommodation coefficients, solid-fluid or fluid-fluid interatomic interactions cause transitions of au and aT near Ar triple-point temperature.
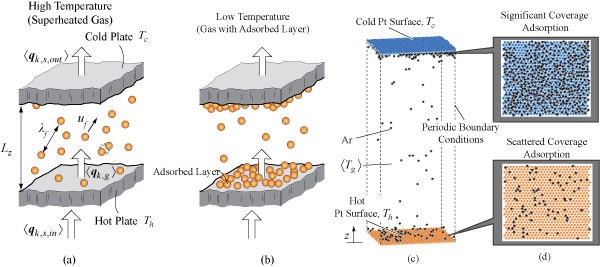
Figure Schematic drawing of heat transfer in gas-filled nanogap both at high (a) and low (b) temperatures. At low temperatures, the adsorbed layers are also shown. (c) 3-D snap shot of simulated Ar atoms (Nf = 600, KnL = 2.1) in the nanogap at = 85 K showing larger adsorption on the top cold surface, and (d) stereo view of the top and bottom surface showing larger adsorption at the cold surface.

